Salinity of Ocean Water

Saltiness of a body of water is salinity. Levels of different salts such as sodium chloride, calcium sulfates and magnesium, and bicarbonates in a water mass are described under the term salinity of water.
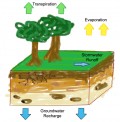
Ocean water is saline to taste as it contains salts dissolved in it. The salinity is measured in terms of the parts of salt dissolved per thousand parts of ocean water. The distribution of salinity in the oceans is not uniform. The salinity in the oceans depends upon a number of factors which lead to dilution or concentration of the saline solution. The temperature and the rate of evaporation and the addition of fresh water by rainfall or large rivers or ice melt are the most important controls of salinity. The average salinity of the open seas is about 32 per thousand. It is higher in the dry tropics where the rate of evaporation is more and the addition of fresh water due to rainfall or large rivers is very little. In the equatorial regions it is slightly lower as these regions receive a large amount of rainfall and many large rivers flow into the seas in these regions. Similarly the salinity is lower in the high latitudes also as the rate of evaporation her is low and there is a large addition of fresh water due to ice melt from the polar and the sub-polar regions. However, the differences in the salinity in the open seas are very limited. They are much greater between the inland seas and the open oceans.
Generally the inland seas in the tropical zone which experience a high rate of evaporation and receive no rivers, e.g. the Red Sea, have a much higher salinity than the open seas. On the other hand the inland seas receiving a lot of river water or ice melt, e.g. the White Sea, have a much lower salinity than the open seas. The most abundant salt in the ocean waters are the sodium chloride, potassium chloride and calcium and magnesium chlorides. Chlorine is the most abundant element dissolved in the ocean water.